Chemo-thromboprophylaxis following Total Hip and Knee Replacements.
Chau Ryan1, Modi Amit1
1Trauma & Orthopaedic Registrar. Department of Trauma & Orthopaedic Surgery, University Hospitals of Leicester, Leicester, United Kingdom LE1 5WW.
Address of Correspondence
Dr. Ryan Chau,
Trauma & Orthopaedic Registrar. Department of Trauma & Orthopaedic Surgery, University Hospitals of Leicester, Leicester, United Kingdom . Le1 5WW.
Email: Ryanchau88@gmail.com
ABSTRACT
Objective: To study the correlation of pre-op muscle strength of Hip Abductors, Quadriceps and Hamstrings to post- op physical function in patients with pre- op and post-op Total Knee Arthroplasty. Materials & Method: Fifty- one subjects were included in the study. Initially the patient was asked to fill WOMAC questionnaire. Then the muscle strength of hip abductors, quadriceps and hamstrings was assessed with hand- held dynamometer. Then the patient was asked to perform timed up and go test.
Result: There was no significant correlation between pre-op muscle strength of Hip Abductors, Quadriceps and Hamstrings to post- op physical function in patients with pre- op and post-op Total Knee Arthroplasty.
Conclusion: Improvement was observed in hip abductors, quadriceps and hamstrings to physical function when compared with pre- op to post- operative data.
Improvement in physical function as measured by WOMAC and TUG test was significant in the studied population. No correlation was observed between pre- op muscle strength of hip abductors, quadriceps and hamstrings to post- operative physical function in patients with Total Knee Arthroplasty.
Key words: Total Knee Arthroplasty, Quadriceps, Hamstrings, Hip Abductors
INTRODUCTION
There has been much developments and changes in the use of chemo-thromboprophylaxis in recent years. This short review aims to give an outline of recent developments and the use of some common chemo-thromboprophylactic agents following total hip replacement (THR) and total knee replacement (TKR) surgery.
Background
Major joint replacement surgery is associated with significant risks of venous thromboembolism (VTE) [1], which includes both deep vein thrombosis (DVT), and pulmonary embolism (PE). Without any form of chemo-thromboprophylaxis following THR and TKR surgery the incidence of proximal DVT and fatal PE are as high as 20-30% and 2-4% respectively [2] With post-operative chemo-thromboprophylaxis using warfarin or low-molecular-weight-heparin (LMWH), these figures are reduced substantially to 5% and 0.1-0.2% respectively [2]. As a result, chemo-thromboprophylactic agents such as LMWH are widely used following THR and TKR.
Duration of prophylaxis
Since LMWH is administered subcutaneously, most patients only receive it for the short period of in-patient hospitalisation. In 1981, Sikorski et al reported an elevated residual risk of VTE from the 4th till the 18th day after THR [3]. Similar findings of delayed VTE following THR and TKR were observed in other studies. Dahl et al proposed that discontinuation of thromboprophylaxis (dalteparin) one week after surgery allows a second wave of coagulation and fibrinolysis activation to occur. Between the 6th and the 35th postoperative day they noticed similar increases in plasma d-dimer, which may indicate possible late DVT formation [4]. Thus, the short duration of thromboprophylaxis post-operatively may be unmasking a delayed hypercoagulability state, and provide false reassurance that anticoagulation is adequate. With this in mind, Dahl et al carried out a double blind randomised controlled trial (RCT) involving 308 patients. All patients received initial thromboprophylaxis with dalteparin (Fragmin) 5000 IU once daily for seven days, and were randomised to receive further thromboprophylaxis or placebo for four more weeks. The study found that the THR group with prolonged thromboprophylaxis had a significantly reduced frequency of DVT (p=0.017) within the extended treatment period. Their subsequent recommendation was that THR patients should have a prolonged thromboprophylaxis for five weeks [5].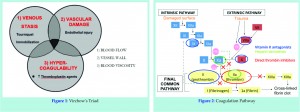
On analysing a larger cohort of patients from California, comprising 19586 THR and 24059 TKR patients over a two year period, White et al found that 76% of VTE in THR patients and 47% of VTE in TKR patients were made only after hospital discharge [6]. The median times of VTE diagnoses were 17 days in THR patients and seven days in TKR patients. The methods and duration of thromboprophylaxis were, however, variable in the studied groups. 95% of the patients received thromboprophylaxis in the form unfractionated heparin or warfarin, with approximately 32% of the patients receiving fours weeks of the latter.
A later Norwegian meta-analysis of 3999 THR and TKR patients by Eikelboom et al provided additional insight, and found that VTE was diagnosed much later post-operatively with a mean of 27 days for THR and 16 days for TKR patients following surgery. They found that four weeks of extended use of prophylaxis, as compared to 7-10 days, did indeed decrease DVT rates significantly, but only in THR patients. However, no significant benefit was found for TKR patients. They also calculated that for every 1000 THRs, 20 DVTs and 1 death were prevented, but with no additional risk of bleeding [7].
With many protocols confining the use of subcutaneous LMWH to in-patients only, and taking into account the increasingly shorter duration of in-hospital stay following elective THR and TKR in recent years (8,9), published evidence would suggest that many institutions are providing an inadequate duration of VTE thromboprophylaxis following THR and TKR. In a more recent study based on data of 6639 THR and 8236 TKR patients from the Global Orthopaedic Registry, Warwick et al reported a mean time to VTE of 21.5 days and 9.7 days for THR and TKR patients respectively. For THR patients 75% of the diagnosis was made after the median time for discharge, and for TKR the figure was 57% [10].
Establishing guidelines
With mounting evidence, a set of guidelines for THR and TKR patients was announced at the 7th American College of Chest Physicians (ACCP) Conference on Antithrombotic and Thrombolytic Therapy in 2004. This recommended an optimal duration of chemo-thromboprophylaxis following TKR for at least ten days, and that patients with THR should be given extended prophylaxis until 28 to 35 days after surgery [11]. The recommendation of a minimal of ten days of post-operative chemo-thromboprophylaxis was given the highest possible 1A grading, indicating that the ACCP has judged these recommendations to be strong and that the benefits of following these recommendations outweigh the risks, burden, and costs.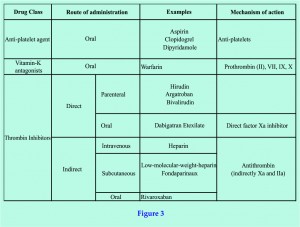
In 2007 the National Institute for Clinical Excellence (NICE) for England and Wales issued their recommendation in the use of LMWH following THR for an extended period of four weeks [12]. In September the following year, a technology appraisal was issued and the use of dabigatran etexilate in the prevention of thromboembolism following THR and TKR surgery was approved [13].
With the new guidelines recommendations, the practicalities in using traditional chemo-prophylactic agent such as LMWH need to be reconsidered. We take a look at some commonly used drugs, as well as recent additions to the market.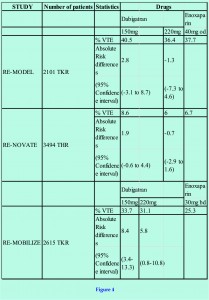
Chemo-thromboprophylaxis
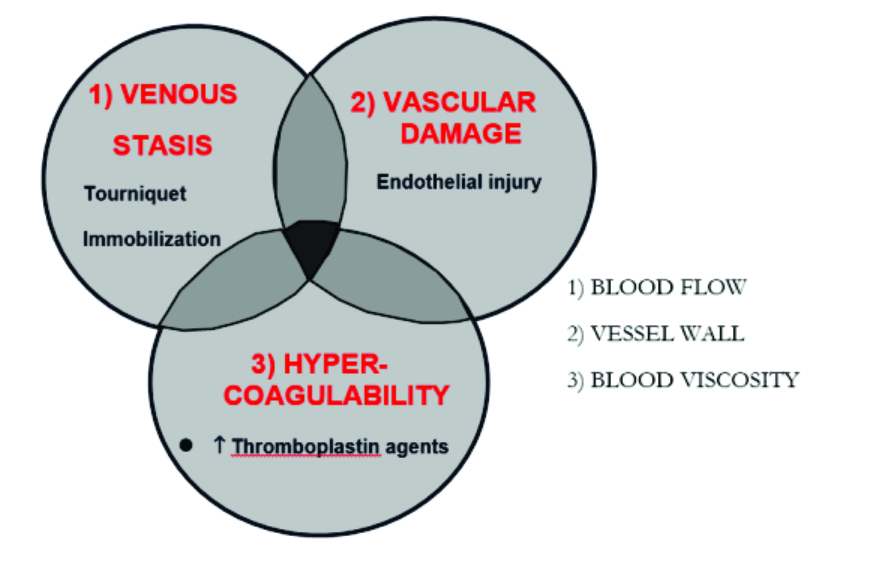
Thromboembolism develops as a result of changes in the constituents to Virchow’s triad (figure 1). In particular, injured blood vessel endothelium interact with coagulation factors from platelets, plasma and damages cells, which activate a cascade that ultimately lead to clots in the form of activated fibrin (figure 2). Physical means of reducing the risk of thromboembolism include adequate hydration, reduced surgery and tourniquet time, and early mobilization. Mechanical devices such as elastic stockings and intermittent compression devices have also been proven useful. More effective still, are pharmacological chemo-thromboprophylactic agents targeting the clotting pathways [1], these may be categorised according to their mechanism of action (figure 3).
ANTIPLATELET AGENTS
Antiplatelets inhibit platelet cyclo-oxygenase, a key enzyme in thromboxane A2 generation. Agents such as aspirin have been used as VTE thromboprophylaxis previously, but like all non-steroidal anti-inflammatory drugs, there is an associated risk of gastro-intestinal bleeding. Post-operative thromboembolic prophylaxis is not as effective as LMWH [1]. In THR patients the risk reduction of DVT is 26% using aspirin compared to 70% with LMWH. In TKR patients the risk reduction is 13% using aspirin compared to 52% with LMWH [14].
VITAMIN K ANTAGONIST
Vitamin K antagonists inhibit the action of reductase enzyme, required for the recycling of oxidized vitamin K, which is needed in the carboxylation of coagulation proteins such as prothrombin and factor VII [figure 2]. A commonly used drug from this group is warfarin, which has a slow onset of action and a relatively long half-life of 25 to 60 hours. Warfarin binds predominantly to albumin and is metabolized extensively in the liver; via the cytochrome P450 pathway. Warfarin has interactions with a wide range of drugs and food, and can cross the placenta to cause fetal bone abnormalities. The narrow therapeutic window, together with genetic variations in metabolism and food interactions, means that close blood level monitoring and frequent dose adjustments are necessary. As such, it is far from the ideal agent for use as chemo-thrombo-prophylaxis following joint replacement surgery.
DIRECT THROMBIN INHIBTORS
Direct thrombin inhibitors have several advantages over their indirect counterpart. They do not bind to plasma proteins or platelet factors, giving them a more predictable anticoagulant response and no risk of HIT.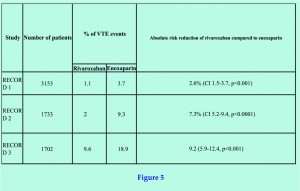
Dabigatran etexilate
Dabigatran etexilate is an oral direct thrombin inhibitor and marketed as a pro-drug under the name of Pradaxa. It is converted into dabigatran in the blood plasma after absorption. Dabigatran level peaks at two hours, and has a half-life of 14 to 17 hours. Eighty percent of dabigatran is excreted by the kidneys, and bypasses the Cytochrome P450 system. The most attractive aspect of the drug is its means of oral administration, and that monitoring of therapeutic levels is not required.
The efficacy and safety of dabigatran etexilate usage were addressed through two double-blind, multicentre, RCT clinical studies (RE-NOVATE for THR, RE-MODEL for TKR) [15,16]. The studies compared controls groups using the current standard, 40mg subcutaneous LMWH enoxaparin once daily, against two oral doses of dabigatran etexilate 220mg, and 150mg. The first dose of dabigatran etexilate was given 6-12 hours after surgery. Treatment was continued for 6-10 days for the TKR group and THR for 28-35 days. Patients were followed up for 12-14 weeks. Primary outcome was VTE events such as asymptomatic DVT; measured by venography at the end of the treatment period, or symptomatic PE within the duration of follow-up. In both studies the majority of VTE were asymptomatic. Both studies demonstrate that dabigatran etexilate does not fare any worse than enoxaparin (figure 4).
For patients in the RE-MODEL study, neither the 150mg (p<0.017) nor the 220mg (p<0.0001) groups faired significantly worse than the group treated with enoxaparin. For the THR 3494 patients in the RE-NOVATE study the non-inferiority result of dabigatran etexilate against enoxaparin was even more pronounced (150mg, p<0.0001, 220mg, p<0.0001, when compared to enoxaparin). Moreover, the study provided reassurance of safety with a comparable incidence of major bleeding events using dabigatran etexilate.
A further phase III study compared similar groups of dabigatran etexilate with 30mg twice-daily dose of enoxaparin, which is used in the North American formulary. The result again indicates that dabigatran etexilate is at least as effective as enoxaparin in thromboprophylaxis [figure 4]. The difference in VTE rate was primarily due the incidence of asymptomatic VTE detected by venography at the end of therapy. Major bleeding events were more common in the enoxaparin group (1.4%) compared to the 150mg and 220mg dabigatran etexilate group (0.6% for both groups), but the difference is not statistically significant.
INDIRECT THROMBIN INHIBITORS
Unfractionated heparin
Heparin increases anti-thrombin activity via indirect actions of factors IIa and Xa (figure 1). Early adoption of heparin proved effective [1]. However, unfractionated heparin gives a short half live of 1-5 hours and requires continuous infusion and regular therapeutic blood monitoring. There are fewer drug interactions but specific complications such as HIT can occur. Naturally, the intravenous route of administration itself renders the use if unfractionated heparin impractical outside hospitals.
LMWH (Low-molecular-weight-heparin)
The discovery of LMWH provided a more convenient way of administration compared to its unfractionated counterpart. The side and potential adverse effects with heparin remains, but it also retains the effectiveness of unfractionated heparin, given via a once daily (or twice in North America) subcutaneous injection. Until recently, heparin has been the most effective pharmacological agent in the prevention of VTE [1]. Although possible, extended administration of LMWH after discharge poses significant inconvenience to patients and may lead to poor compliance [9].
Rivaroxiban
Rivaroxaban is the first available orally active direct factor Xa inhibitor. Thrombin formation is reduced via its inhibitory action. Rivaroxaban is marketed as Xarelto and has been approved for use in EU and Canada for the prevention of VTE in adults undergoing THR or TKR.
Like dabigatran, Rivaroxaban underwent multiple doubled-blind, randomized control studies (RECORD 1, 2 and 3), comprising a pooled data of more than 9500 patients [18,19,20]. The trials compared a once daily dose of 10mg oral rivaroxaban with 40mg subcutaneous enoxaparin (Figure 5).
In RECORD 1, 2 and 3, the first dose of rivaroxaban was given six to eight hours after wound closure. In contrast, the first dose of enoxaparin was given 12 hours prior to surgery with the second dose six to eight hours after wound closure. In RECORD 1 and 2, rivaroxaban treatment was continued for up to 39 days, whereas enoxaparin was stopped after 14 days. In RECORD 3 both drugs were used for 10-14 days only. Bilateral venography was performed after the last dose, with follow up for a further 30-35 days afterwards.
The outcome measure was a combination of asymptomatic DVT, or symptomatic DVT or PE. Overall, rivaroxaban treated patients had major VTE of 0.2-1% compared to 2-5.1% in the enoxaparin treated cohort. Enoxaparin showed an absolute risk reduction of 2.6% (confidence interval 1.5-3.7), with a relative risk ratio of approx 50-70%. In other words, rivaroxaban provided a significantly more effective chemo-thromboprophylaxis following THR and TKR than even the current standard using enoxaparin.
There was no significant difference in cardiovascular events, liver enzyme derangements or major bleeding events between the groups in the studies, although surgical site bleeding was not assessed unless it resulted in further re-operation or fatality.
Similar to the dabigatran etexilate series, there was a further RECORD 4 study, which compared rivaroxaban with 30mg twice daily of subcutaneous enoxaparin, as used in the North American formulary [21]. The results indicate that oral rivaroxaban 10 mg once daily for 10 to14 days was significantly superior to enoxaparin in preventing VTE following total knee arthroplasty. VTE occurs in 67 (6·9%) of 965 patients given rivaroxaban and in 97 (10·1%) of 959 given enoxaparin (absolute risk reduction 3·19%, 95% CI 0·71–5·67; p=0·0118). Ten (0·7%) patients in the rivaroxaban group had major bleeding events compared to four (0·3%) in the enoxaparin group, but the difference is insignificant (p=0·1096).
The Future
The importance of thromboprophylaxis for VTE cannot be overstated. From 1997-2007, there were 776 claims of alleged failure to use prophylaxis, which lead to DVT and death in England and Wales. For 633 patients who died of pulmonary embolus, the claimant alleges that the hospital provided insufficient prevention for VTE. Overall, £68 million were paid out for claims relating to VTE. Whilst not all of this is directly relating to orthopaedic and trauma surgery, a significant risk is imposed onto patients undergoing major emergency or elective trauma and orthopaedic surgery, with THR and TKR being the most common surgery in the latter group.
The current healthcare guidelines have highlighted the inadequacy of chemo-thromboprophylaxis, and the extended regime is a welcomed recommendation. However, traditional anticoagulants such as LMWH are inconvenient for home usage and poor compliance is likely with prolonged use. Anticoagulants such as dabigatran or rivaroxaban may provide a more practical solution.
There are still other outstanding issues to be addressed, such as the timing for the first dose of the anticoagulant, and associated effects such as oozing wounds and bleeding events. Dabigatran etexilate and rivaroxaban given in the trials described were administered 4 to 8 hours post-operatively. There is some suggestion that peri-operative, or even pre-operative administration of LMWH may provide more effective thromboprophylaxis, although there is an increased risk of bleeding [22,23].
Without doubt, clinical trials of novel anticoagulants will continue, and its adoption in widespread usage will be largely dependent on efficacy and cost. We are likely to see continual and parallel developments in both Xa inhibitors and direct thrombin inhibitors, whilst other newer drugs, targeting the mechanism of actions of coagulation factors (Va, VIIa and IXa) will proceed to more clinical trials in the near future..
References
1. Geerts WH, Pineo GF, Heit JA, Bergqvist D, Lassen MR, Colwell CW, Ray JG. Prevention of venous thromboembolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest. 2004 Sep;126(3 Suppl):338S-400S.
2. Stein PD, Hull RD, Matta F, Yaekoub AY. Early discharge of patients with venous thromboembolism: implications regarding therapy. Clin Appl Thromb Hemost. 2010 Apr;16(2):141-5. Epub 2009 Oct 14.
3. Eikelboom JW, Quinlan DJ, Douketis JD. Extended-duration prophylaxis against venous thromboembolism after total hip or knee replacement: a meta-analysis of the randomised trials. Lancet. 2001 Jul 7;358(9275):9-15.
4. Hull RD, Pineo GF, Stein PD, Mah AF, MacIsaac SM, Dahl OE, Butcher M, Brant RF, Ghali WA, Bergqvist D, Raskob GE. Extended out-of-hospital low-molecular-weight heparin prophylaxis against deep venous thrombosis in patients after elective hip arthroplasty: a systematic review. Ann Intern Med. 2001 Nov 20;135(10):858-69
5. Prandoni P, Bruchi O, Sabbion P, Tanduo C, Scudeller A, Sardella C, Errigo G, Pietrobelli F, Maso G, Girolami A. Prolonged thromboprophylaxis with oral anticoagulants after total hip arthroplasty: a prospective controlled randomized study. Arch Intern Med. 2002 Sep 23;162(17):1966-71.
6. Geerts WH, Bergqvist D, Pineo GF, Heit JA, Samama CM, Lassen MR, Colwell CW. Prevention of venous thromboembolism: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest. 2008 Jun;133(6 Suppl):381S-453S.
7. Eriksson BI, Dahl OE, Ahnfelt L, Kälebo P, Stangier J, Nehmiz G, Hermansson K, Kohlbrenner V. Dose escalating safety study of a new oral direct thrombin inhibitor, dabigatran etexilate, in patients undergoing total hip replacement: BISTRO I. J Thromb Haemost. 2004 Sep;2(9):1573-80.
8. Eriksson BI, Dahl OE, Büller HR, Hettiarachchi R, Rosencher N, Bravo ML, Ahnfelt L, Piovella F, Stangier J, Kälebo P, Reilly P; BISTRO II Study Group. A new oral direct thrombin inhibitor, dabigatran etexilate, compared with enoxaparin for prevention of thromboembolic events following total hip or knee replacement: the BISTRO II randomized trial. J Thromb Haemost. 2005 Jan;3(1):103-11.
9. Eriksson BI, Dahl OE, Rosencher N, Kurth AA, van Dijk CN, Frostick SP, Prins MH, Hettiarachchi R, Hantel S, Schnee J, Büller HR; RE-NOVATE Study Group. Dabigatran etexilate versus enoxaparin for prevention of venous thromboembolism after total hip replacement: a randomised, double-blind, non-inferiority trial. Lancet. 2007 Sep 15;370(9591):949-56.
10. Schulman S, Kearon C, Kakkar AK, Mismetti P, Schellong S, Eriksson H, Baanstra D, Schnee J, Goldhaber SZ; RE-COVER Study Group. Dabigatran versus warfarin in the treatment of acute venous thromboembolism. N Engl J Med. 2009 Dec 10;361(24):2342-52.
| How to Cite: Ryan C, Amit M. Chemo-Thromboprophylaxis following total Hip and Knee Replacements. Journal of Orthopaedic and Rehabilitation 2015 Jult-Sep; 1(2): 39-42 |
[Full Text HTML] [Full Text PDF]
